ISO 17025 Certification In Saint Louis
ISO 17025 certification In Saint Louis, Factocert is one of the leading ISO consultants in Senegal. We provide the best ISO 17025 certification auditors in Saint Louis and other major cities with the service of consultation, implementation, documentation, certification, audit, and other related services all across the world at an affordable cost.
ISO 17025 certification in Saint Louis aids the organizations which contain laboratories to possess a continual top quality system so that the organization can observe the improvement and consistency. It is one of the internationally recognized standards that specifies the overall requirements for testing and calibration of laboratories. The prerequisites are made in such a way they are globally recognized based on their certificate.
5 Steps to get ISO 17025 certification in Saint Louis
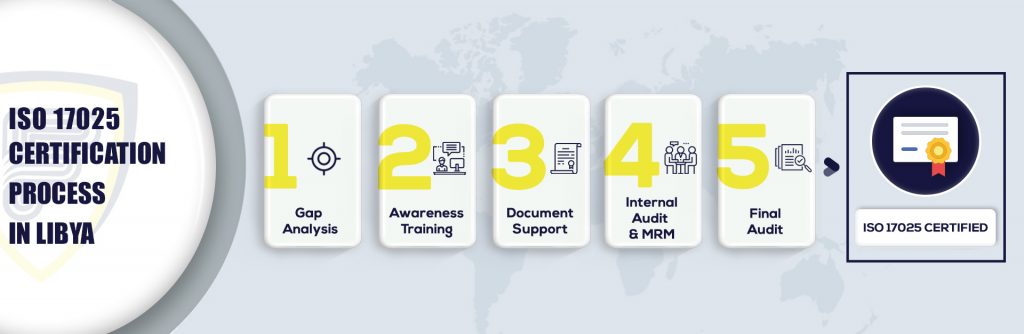

What’s the ISO 17025 Certification in Saint Louis means?
ISO 17025 certification in Saint Louis covers calibration and testing using standard methods, non-standard strategies and laboratory improved methods. The organizations comprising labs, which wants to build up their management system for technical, administrative and quality surgeries, can proceed for ISO/IEC 17025 standard.
The demands of ISO/IEC 17025 are categorized into two kinds:
- Technical demands: Technical requirements of ISO/IEC 17025 largely relates to calibration and testing processes like technical personnel, testing and calibration procedures, equipment’s, sampling, measurement of traceability, testing and calibration items handling, quality assurance results and other reporting results related to labs.
- Management requirements.:Management requirements are primarily centred on management and update of documents organizations observation optimization of the quality management system and evaluation of the contracts
Why ISO/IEC 17025 has been revised?
It is necessary to monitor the changes and adjustments from the standard and prepare them to fulfil up the laboratory requirements.
The ISO 17025 standard was revised to boost the data inaccuracy and error cases. The standard has had just two revisions until now, the first one was revised in the year 2005, and recently it had been revised in the calendar year 2017 to match up with the up-to-date sector demands and to follow along with the ISO 9001 standard needs carefully as possible.
The revision of every international standard was performed to accommodate the most recent vital changes in the organization and work practices. Coming to ISO/IEC 17025 standard, the newest changes includes a new chapter on controlling of non-conformity of testing and calibration function, risk-based believing, the new terminologies to cover calibration, testing and sampling and incorporation of personal computer systems and controlling the data.
The approach followed in ISO/IEC 17025 standard currently matches with the newest standards requirements like ISO 9001 quality management system, ISO 15189 which specifies the demands of medical laboratories and ISO 17021 — 1 which define the requirements for auditing and certification bodies.


Henceforth, There is ISO 17025 Certification in Saint Louis standard which is one such beautiful accreditation that includes requirements for testing labs. It aids the labs to show that they’re technically competent and be sure the results are very accurate.
To claim the technical competency of analyzing Calibration Laboratories ISO 17025 Certification in Saint Louis standard comprises technical and management requirements and getting updated with the standard proves that the results are extremely precise and consistent and therefore are generated based on the protocols of this industry standard
Advantages of ISO 17025 Certification in Saint Louis
Implementation of ISO 17025 certification in Saint Louis will benefit your organization by enabling its best possible profile.
- By establishing policies from the laboratories, it is going to help determine the best roles and the actions will empower improvisation from the system. With the decline in error with the help of an appropriate risk management system, the organization can perform more jobs with greater responsibilities.
- By understanding the client requirements, it is easier for the organization to develop a successful path for your laboratory advancement.
- The aggressive mindset of celebrities at the laboratories will promote the expert degree of one of the departments. This will enable the organization to have more appropriate outcomes.
These are the few Advantages of ISO 17025 certification in Saint Louis.
Our services
ISO 9001 sets out the standards for a quality management system. It can be used by any organization, big or small, irrespective of its field of activity. In actuality, there are over one million companies and organizations in over 170 countries certified to ISO 9001.
ISO 9001 wants to show its ability to consistently provide products and services which meet customer and applicable statutory and regulatory demands, and also this standard aims to enhance customer satisfaction through the effective application of the system, including procedures for improvement of the system and the assurance of conformity to customer and applicable legal and regulatory requirements.
ISO 14001 lays out the criteria for an environmental management system and can be licensed to. It maps out a frame that a company or organization can follow to establish a successful environmental management system.
ISO 14001 helps an organization achieve the planned consequences of its environmental management system, which offer value for the environment, the organization itself and interested parties.
ISO 45001 specifies requirements for an occupational health and safety (OH&S) management system, and advises on its use, to enable organizations to provide safe and healthy workplaces by preventing work-related harm and ill health, as well as by proactively improving its OH&S performance.
Many people died from work-related injuries or diseases. That is the reason an ISO committee of occupational health & safety experts set to work to develop an International Standard using the capability to save workers in the office thus ISO introduced ISO 45001 standard.
ISO 22000 sets out the requirements for a food safety management system. It maps out what an organization has to do to demonstrate its ability to control food safety risks to be able to make sure that food is safe. It can be employed by any organization irrespective of its dimensions or position from the food chain.
ISO 31000, Risk management — Guidelines, provides principles, a framework and a method for handling risk. It can be used by any organization irrespective of its dimensions, activity or sector. ISO 31000 provides a level of reassurance concerning financial resilience, professional standing and environmental and safety effects.
ISO 31000 can be utilized throughout the life of the organization and can be applied to any action, such as decision-making at all levels.
ISO 10002 standard is related to Customer satisfaction and Provides Guidelines for complaints handling in organizations.
This Standard guides the process of complaints handling associated with products in an organization, including planning, design, performance, maintenance, and improvement. The complaints-handling procedure described is suitable for use among the procedures of an overall quality management system.
ISO/IEC 27001 standard would be the work of joint ISO and IEC (International Electrotechnical Commission) specialized analysis, Information security, cybersecurity and privacy security.
ISO/IEC 27001 is broadly known, providing requirements for an information security management system (ISMS), although there are over a dozen standards in the ISO/IEC 27000 family. Utilizing them empowers organizations of any kind to manage the security of assets like financial information, intellectual property, employee information or information secured by third parties.
By setting, implementing, maintaining, and information security management system inside the organization, ISO/IEC 27001 certification will help in the evaluation and therapy for information security risks.
ISO 22301 is a Business continuity management system. This will ensure the protection of society from, and in reaction to, incidents, emergencies and disasters brought on by intentional and unintentional human actions, natural hazards and technological failures.
This standard specifies requirements to implement, preserve and improve a management system to protect against, reduce the chance of the incidence of, and prepare for, respond to and recover from disruptions when they arise.
ISO 50001 designed to encourage organizations in all industries, this ISO standard provides a sensible means to better energy use, through the development of an energy management system (EnMS).
This is based on the management system model of constant advancement also used for other renowned standards such as ISO 9001 or ISO 14001. This also makes it much easier for organizations to integrate energy management into their overall efforts to increase quality and environmental management.
ISO 29990: This standard specifies Basic prerequisites for providers of learning services within non-formal education and training.
ISO 29990 is also a quality management system standard for providers of education and training services. ISO 29990 offers a unified model for a quality and skilled mode of performance, as well as a frequent reference point for both learning service providers (LSPs) and their customers for the conceptualization, development, and delivery of the designated program.
ISO 13485 specifies requirements for a quality management system for the medical devices at which an organization needs to demonstrate its ability to provide medical devices.
Such organizations may be involved in one or more stages of their life-cycle, such as design and development, manufacturing, storage and distribution, installation, or servicing of a medical apparatus and layout and development or supply of associated activities. ISO 13485:2016 can also be used by providers or outside parties that provide merchandise, such as quality management system-related services to these organizations.
ISO 13485 is designed to be utilized by organizations involved in the design, manufacturing, installation and servicing of medical devices and related services. In addition, it can be utilized by internal and external parties, such as certification systems, to help them with their auditing processes.
Other services
CE Mark The Conformitè Europëenne (CE) Mark is defined as the European Union’s (EU) mandatory conformity signalling for regulating the goods marketed in the European Economic Area (EEA) since 1985. The CE marking represents a manufacturer’s declaration that goods comply with the EU’s New Approach Directives. These directives not just apply to goods within the EU but also for products that are produced in or made to be sold at the EEA. This makes the CE marking recognizable worldwide even to those unfamiliar with the EEA.
There’s an EU demand that products not in conformity with the provisions of the directives Aren’t Permitted to circulate in the lands of the member nations.
CE marking doesn’t offer any specific information to the consumer. It’s not a quality assurance declaration, it doesn’t demonstrate evidence of third-party testing, and it should not be mistaken with any certification mark of this type issued by international or European informed evaluation bodies.
HACCP Hazard analysis and critical control points, or HACCP is a systematic preventative approach to food safety from the biological, compound, physical dangers and more recently radiological risks in manufacturing processes which could cause the final product to be unsafe and layouts measures to reduce these risks to a secure level. In this manner, HACCP attempts to steer clear of risks instead of trying to inspect finished products for the effects of these threats.
The HACCP system can be used in whatsoever phase of a food chain, from food production and preparation processes including packaging, supply, etc.
HACCP has been acknowledged internationally as a reasonable instrument for adapting conventional inspection methods into some contemporary, science-based, food safety systems. According to risk assessment, HACCP plans enable both businesses and authorities to devote their resources efficiently by building and auditing protected food manufacturing practices.
The OECD Principles of Good Laboratory Practice (GLP) guarantee the creation of high quality and reliable test information related to the safety of industrial chemical substances and preparations.
Vulnerability Assessment and Penetration Testing (VAPT) are equally security services that are devoted to identifying vulnerabilities in the system, server and system infrastructure. The two services serve different functions and are completed to attain different but complementary goals.
Halal is a term designating any item or an action that’s permissible to use or participate in, based on Islamic law. It is the opposite of haram. The term is used to designate food viewed as permissible according to Islamic law. Halal is an Arabic word meaning lawful or permitted. Concerning food, it’s the dietary standard, as prescribed in the Qur’an.
These terms are generally used concerning food products, meat products, cosmetics, healthcare products, pharmaceuticals, food ingredients, and food contact substances. When many items are halal or haram, there are a few items that aren’t clear. Further information is required to categorise them as halal or haram.
Good Manufacturing Practice (GMP) is a system for ensuring the products are consistently produced and controlled according to quality standards. It’s intended to decrease the risks involved in any pharmaceutical production that must not be eliminated through testing the final product.
The SA8000 Standard is the world’s leading social certification application. Even the SA8000 Standard and Certification System offer a framework for organizations of all types, in virtually any business, and in almost any country to conduct business in a way that’s reasonable and decent for workers and also to show their adherence to the highest social standards. Created by SAI in 1997 as the first credible societal certification, it has led the market for more than 20 years.
Capability Maturity Model Integration (CMMI) is a process level enhancement training and appraisal program. Administered by the CMMI Institute, it was developed at Carnegie Mellon University (CMU). It is required by several U.S. Government contracts, especially in program development.
Why Choose Factocert for ISO 17025 certification in Saint Louis?
Factocert is one of the leading Consulting firms which offer fantastic services by using the international standards based on the government set up by the committee of the International Organization for standardization. It is composed of experienced experience who have licensed many Organizations and supplied them with complete solutions.
It is challenging for the representative of the Organization to deal with these implementation processes due to these reasons the Organization has to decide on a Consulting firm like Factocert.
How to get ISO 17025 Certification in Saint Louis?
Wondering How to get ISO 17025 certification in Saint Louis! We will help you with your certification queries and requirements, just drop an email here at contact@factocert.com and even get a free quote from us for ISO 17025 cost in Saint Louis. We provide the best ISO 17025 consultants in Saint Louis to help companies to go through the guidelines set by the international organization for standardization for achieving ISO 17025 Certification in Saint Louis and We always make sure that the Customers are fulfilled with our services along with the means of consulting, If You’d like to know more about our consultation methodology please do visit our site www.factocert.com will be happy to help you.
For more information visit ISO 17025 certification in Saint Louis
Our Services
Our Clients










































ISO 17025 audit services in Saint Louis are essential because if you’re facing the ISO 17025 audit in Saint Louis you have to be very careful about every parameter in your organization but when you join hands with Factocert. We as an ISO 17025 Consultancy Service provider in Saint Louis, are tagged up with so many different ISO 17025 certification bodies in Saint Louis.
Benefits of ISO 17025 Certification in Saint Louis
You will have the edge over your list of ISO certified companies in Saint Louis
Marketing becomes very easy when you have ISO 17025 certification in Saint Louis tagged on to your name and which will help you to get into a global market and be a global player.
Employee satisfaction rates increases which are directly proportional to your employee retention and by retaining your critical employees you will have higher stability as a company
A government will recognize you for having such ISO 17025 certification in Saint Louis
You will be automatically qualifying for any tenders because most of the companies require you to certify for ISO 17025 certification in Saint Louis for participating in the tender
Customer satisfaction rates will go high, and you will not have to face any more consequences from your customers or your vendors
Are you looking for
ISO 17025 Certification Consultants in Saint Louis
What else are you waiting for the only step you have to take care of is getting in touch with us, and we would take the best care, and in no time you would be certified for relevant ISO 17025 certification in Saint Louis you prefer. Most thing companies worry about is ISO 17025 service cost in Saint Louis but let us just tell you that ISO 17025 cost in Saint Louis is not what you should be thinking of because when this certification can give a boost to your organization’s process. We will make sure that the cost of ISO 17025 Certification in Saint Louis is as minimal as possible.
To get you ISO 17025 Certification Services in Saint Louis than we assure you 100% guarantee results and we ensure that you will definitely be certified because have 100% success rates to date in getting our customers certified. So get in touch with us as early as possible and get your ISO 17025 certification in Saint Louis at the earliest.
Mail us at contact@factocert.com for quick assistance.




















