ISO 13485 Certification In Kuwait
ISO 13485 Certification in Kuwait
ISO 13485 Certification In Kuwait, Factocert is one of the leading ISO 13485 Certification providers in Kuwait. We provide ISO Consultant services in Mangaf Al Jahra, Salmiya, Sabah Al Salem, Al Farwaniyah, and other major cities.
ISO 13485 certification in Kuwait is one of the globally recognized standards for Quality Management systems concerning the design and the manufacturing of medical devices. ISO 13485 certification services in Kuwait City, medical devices have the requirements for regulatory purposes. ISO 13485 registration services in Al Ahmadi are beneficial for a comprehensive quality management system for the organization that deals with the design and manufacture of medical devices.
ISO 13485 Certification in Kuwait reminds a stand-alone document but organized with ISO 9001, which globally recognized as a quality management system (QMS). However, the quality management system that is ISO 9001 standard specifies the requirements for the organization to demonstrate continual improvement wherein ISO 13485 audit services in Kuwait define quality management system with practical implementation and maintenance of medical devices. ISO 13485 certification in Kuwait often states that compliance in achieving the regulatory requirements concerning medical devices.
What are the steps to get ISO Certification in Kuwait?
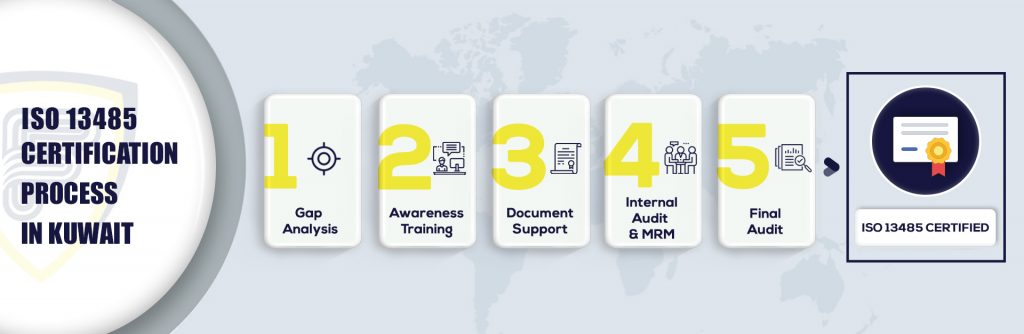

More about ISO 13485 Certification in Kuwait:
ISO 13485 consultants in Kuwait defines that the requirement of the medical devices when it comes to manufacturing industries must have incorporated documentation. Here will help the organization to identify the appropriate management system that has achieved compliance in medical device industries. ISO 9001 Certification in Kuwait standard emphasizes more on the continual improvement in each process ISO 13485 standard particularly emphasize on meeting regulatory and customer requirements on time.
ISO 13485 Certification in Kuwait also takes care of risk management maintaining effective methods in many other criteria where the manufacturer of medical devices organization has safe design, manufacture, and distribution concerned. We provide the best quote for ISO 13485 certification cost in Kuwait. ISO has three official languages such as English, French, and Russian. ISO, as published more than 20000 different international standards and ISO 13485 standard, is one of them. ISO 13485 Certification in Kuwait helps the organization in meeting legislative regulator and customer requirements. The international standard will also help the manufacturer of medical devices to have a safe and global exchange of business. Implementation of the ISO 13485 Certification in Kuwait will help the organization to have a global market.
Few important facts on ISO 13485 Certification in Kuwait:
The organizations which are into the medical field will have the ambiguity of specifying specific units when it comes to designing a medical device, but ISO 13485 Certification in Kuwait will help in solving the complexity from country to country. The primary benefit for the company from the ISO 13485 certification in Kuwait is process improvement, quality service, best manufacturing practices, and excellent management assistance.
In the pharmaceutical industry, there is a wide variety of methods or tools are used, ranging from essential hand tools to complex computerized machines. Every small device, such as a stethoscope to an MRI scanning machine, should have durability and reliable function. Any minor glitches in the instrument such as life-supporting tools our lungs ventilator can cause a humongous loss. Most of the medical devices which are control by software program type such as CT scanners should have an experienced person in charge.


How PDCA cycle helps – ISO 13485 certification in Kuwait?
ISO 13485 certification in Kuwait City ensures that the organization should have a set of policies, procedures to design for the manufacture of research devices for the medical industries.
The implementation of ISO 13485 consultant services in Kuwait involves many activities such as the PDCA cycle, the primary management handling process.
With the help of this PDCA cycle, the management can have a controlled and continuous improvement in each process, which will benefit both in the long term and short. In any medical device industry, it is a human-made tree step to have a controlled quality management system. The required regulator is must place in each department to achieve an active medical device. ISO 13485 Certification in Kuwait focuses on customer satisfaction and regulatory requirements.
our services:
ISO 9001 sets out the criteria for a quality Management system and is the only standard in the family that can be licensed to (although this isn’t a necessity ). It may be employed by almost any organization, large or small, regardless of its field of activity. There are over one million businesses and organizations in over 170 nations certified to ISO 9001.
Based On the International Labour Organization, more than 7 600 individuals die from work-related accidents or diseases every single moment. That’s the reason an ISO committee of occupational health & safety experts place to work to develop an International Standard with the capability to save nearly three million lives each year.
Structured in a similar way to other ISO management systems, the approach will be familiar to consumers of standards such as ISO 14001 or ISO 9001. ISO 45001 builds on the achievement of previously international standards in this field including OHSAS 18001, the International Labour Organization’s ILO-OSH Guidelines, various federal standards, and the ILO’s international labor standards and standards
ISO/IEC 17025 enables laboratories to show That they operate competently and create valid benefits, thereby promoting confidence in their work both nationwide and around the globe.
Additionally, it helps ease collaboration Between laboratories and other bodies by producing wider acceptance of results between countries. Test reports and certificates may be accepted from 1 nation to another without the need for further testing, which, in turn, improves international trade.
The consequences of unsafe food can be severe. ISO’s food safety management standards assist organizations to identify and control food safety hazards, at precisely the same time as working together with other ISO management standards, including ISO 9001. Applicable to all sorts of producers, ISO 22000 supplies a coating of reassurance inside the worldwide food distribution chain, helping products cross borders and bringing folks food that they can trust.
ISO/IEC 27001 is widely known, supplying requirements for the information security management system (ISMS), even although there are over a dozen standards in the ISO/IEC 27000 family. Utilizing them empowers organizations of any type to handle the security of assets like financial information, intellectual property, employee information, or information entrusted by third parties.
It requirements for the organization to set up, implement, maintain and constantly improve a service management system (SMS). The requirements specified within this document include the preparation, design, transition, delivery, and development of services to satisfy up the service demands and provide value.
ISO 31000, Risk management — Strategies, Supplies principles, a framework, and a process for managing risk. It may be used by any organization regardless of its size, action, or sector.
Employing ISO 31000 may help Organizations increase the chance of achieving goals, improve the identification of opportunities and risks and effectively allocate and use funds for risk treatment.
However, ISO 31000 Cannot be used For certification purposes but can guide external or internal audit programs. Organizations using it can evaluate their risk management practices using an internationally recognized benchmark, supplying sound fundamentals for effective management and corporate governance.
This standard offers guidelines for the Practice of Complaints handling related to products and services within an organization, such as planning, design, creation, operation, upkeep, and improvement. The complaints-handling process described is suitable for use as one of the procedures of an overall quality management system.
This standard specifies requirements to execute, Maintain and enhance a management system to protect against, reduce the probability of the incidence of, prepare for, respond to and recover from disruptions if they arise.
The requirements specified within this Document are generic and intended to be relevant to most organizations, or parts thereof, regardless of type, size, and nature of the organization. The level of use of the requirements is contingent on the organization’s working environment and sophistication.
ISO 14001 lays out the criteria for an environmental management system and may be accredited. It maps out a framework that a business or organization can follow to set up a successful environmental management system.
Designed for any type of organization, Regardless of its activity or sector, it may assure the business management and employees in addition to external stakeholders that environmental impact has been measured and enhanced.
For organizations committed to addressing their effect, preserving resources, and improving the bottom line through effective energy management, we developed ISO 50001. Designed to support organizations in all industries, this ISO standard provides a sensible way to improve energy use, through the development of an energy management system (EnMS).
The objective Of ISO 29990 will be to offer a standard model for quality expert practice and performance, and a frequent benchmark for teach service providers (LSPs) and their clients in the design, development, and delivery of non-formal education, training, and advancement. This International Standard uses the term”learning services” rather than”training” to be able to promote a focus on the student and the results of the procedure and to highlight the full selection of alternatives available for delivering coaching services.
The Conformitè Europëenne (CE) Mark is defined as The European Union’s (EU) mandatory conformity indicating for regulating the goods sold within the European Economic Area (EEA) as of 1985.
The CE marking represents a manufacturer’s declaration that goods comply with the EU’s New Approach Directives. These directives not just apply to products inside the EU but also for products that are manufactured in or designed to be marketed at the EEA. This produces the CE marking recognizable worldwide even to people unfamiliar with the EEA.
Halal signifies”permissible” In Arabic and refers to food items that are permissible to absorb under Sharia law, whereas haram (lit. “criminal”) describes any material not permitted to absorb.
According to the Australian Food and Grocery Council, halal foods should be”free from any the substance is extracted or taken from a haram creature or ingredient (e.g. pigs, dogs, carnivorous animals, animals not slaughtered according to Islamic rites); [be] produced, processed, manufactured, and/or preserved by using utensils, gear, and/or machinery that’s been cleaned by Islamic law (e.g. not cleaned with alcohol); and [be] free in contact with, or being close to, a haram substance during preparation, manufacture, processing, and storage (e.g. blood, alcohol, poisonous and magnifying plants and bugs such as worms and cockroaches).”
Is a Quality organization of the Government of Kerala Providing cost-effective & professional assessment, audit, and certification of Hazard Analysis Critical Control Point (HACCP) System for the food industry and other food-related sellers.
What is HACCP?
HACCP (Hazard Analysis Critical Control Point) is an internationally accepted technique for preventing microbiological, chemical, and physical contamination across the food supply chain.
The HACCP method does this by identifying the risks, establishing critical control points, setting critical limits, and ensuring control steps are validated, confirmed, and tracked before implementation.
The successful implementation of HACCP Will improves the capability of businesses to protect and enhance brands and private labels, promote consumer confidence, and conform to regulatory and market conditions.
GMP certification is essential. Good Manufacturing Practice is nothing but measures that are taken to have a controlled practice based on quality standards.
Good manufacturing practice (GMP) is a system for ensuring that products are consistently produced and controlled by quality standards. It is intended to decrease the risks involved in any pharmaceutical production that may not be eliminated throughout testing the final product.
The key risks are unexpected contamination of merchandise, causing damage to health or even death; incorrect tags on containers, which could mean that patients get the wrong medicine; inadequate or too much active component, resulting in ineffective treatment or adverse effects. GMP covers all elements of production; from the beginning materials, premises and equipment to this training and personal hygiene of employees.
Detailed, written procedures are essential for each process that could affect the quality of the final product. There have to be systems to provide documented evidence that proper procedures are always followed at each step of the manufacturing process – every moment a product is created. WHO has established detailed guidelines for good manufacturing practices. Most nations have formulated their particular needs for GMP based on WHO GMP. Others also have harmonized their needs, for example in the Association of South-East Asian Nations (ASEAN), in the European Union, and through the Pharmaceutical Inspection Convention.
The SA8000 Standard is that the World’s top social certification application. Even the SA8000 Standard and Certification System supply a framework for organizations of all types, in virtually any market, and in almost any country to conduct business in a means that’s fair and adequate for workers and to show their adherence to their greatest social standards. Created by SAI in 1997 as the first respectable societal certification, it has led the business for more than 20 years.
Vulnerability assessment and penetration testing Are the two sorts of tests comprising different strength and total vulnerability assessment. Any cybersecurity exposures could be recognized with the help of these two security testing methods.
is a process-level improvement training and Evaluation program. Administered from the CMMI Institute, a subsidiary of ISACA, it had been developed at Carnegie Mellon University (CMU). It is demanded by most U.S. Government Contracts, particularly in software development. CMU asserts CMMI may be used to guide process improvement across a project, branch, or whole organization.
CMMI defines The subsequent maturity levels for processes: Initial, Managed, Defined, Quantitatively Managed, and Optimizing. Version 2.0 was published in 2018 (Version 1.3 was published in 2010, and it is now the reference model for the Remaining information within this wiki essay ). CMMI is registered in the U.S. Patent and Trademark Office by CMU.
Advantages of ISO 13485 Certification in Kuwait:
- ISO 13485 standard demonstrates competency along with the regulatory and legal requirements of pharmaceutical industries.
- ISO 13485 Certification in Kuwait Here will help to have consistency in each process, providing excellent results which are safe and effective for medical devices.
- A well-organized management system that will ensure that risk must address with an effective factor.
- Improve process and observe significant efficiencies required after the implementation of ISO 13485 certification in Kuwait.
- The organization which is having a sound management system will also gain competency in the Marketplace among the competitors.
- Implementation of the ISO 13485 Certification in Kuwait standard will have a more significant benefit for both the customer and the company. The benefits of ISO 13485 Certification in Kuwait are enormous.
- Requirements in this particular standard will improve the condition of the infrastructure, particularly when it comes to the production team concerning device handling matters.
- Medical devices must have an ideology concerning sterile equipment.
- ISO 13485 Certification in Kuwait standards helps to have a regulatory requirement with increased alignment concerning standard aspects.
Why ISO 13485 Certification is important:
ISO 13485 Certification in Kuwait will also help to pass many criteria with stable medical conditions in the devices concerning having excellent manufacture. The organization should have a productive team to design and develop a medical device that meets standard requirements. This unit should also have a sub of a group of involving when it comes to repairing these devices. ISO 13485 standard ensures that the organization should take care of supplying enough raw materials and other components which are necessary for manufacturing a proper functional medical device. ISO 13485 registrations in Kuwait also assist the organization to have a good distribution practice of medical devices.
ISO 13485 standard ensures that the organization provides enough care about taking care of medical devices from the beginning till the point where it reaches the customer and going further. The organization should have robust planning for the verification, transfer, and maintenance of these devices. Know more about ISO 13485 cost in Kuwait by sending an inquiry to contact@factocert.com.
For More Information: ISO 13485 Certification in Kuwait
Our Services
Our Clients










































ISO 13485 audit services in Kuwait are essential because if you’re facing the ISO 13485 audit in Kuwait you have to be very careful about every parameter in your organization but when you join hands with Factocert. We as an ISO 13485 Consultancy Service provider in Kuwait, are tagged up with so many different ISO 13485 certification bodies in Kuwait.
Benefits ISO 13485 Certification in Kuwait
You will have the edge over your list of ISO certified companies in Kuwait.
Marketing becomes very easy when you have certification tagged on to your name and which will help you to get into a global market and be a global player.
Employee satisfaction rates increases which are directly proportional to your employee retention and by retaining your critical employees you will have higher stability as a company
A government will recognize you for having such ISO 13485 certification in Kuwait
You will be automatically qualifying for any tenders because most of the companies require you to certify for ISO 13485 Certification in Kuwait for participating in tender
Customer satisfaction rates will go high, and you will not have to face any more consequences from your customers or your vendors
Are you looking for
ISO 13485 Certification Consultants in Kuwait
What else are you waiting for the only step you have to take care about is getting in touch with us, and we would take the best care, and in no time you would be certified for relevant ISO 13485 certification in Kuwait you prefer. Most thing companies worry about is ISO service cost in Kuwait but let us just tell you that ISO cost in Kuwait is not what you should be thinking of because when this certification can give a boost to your organizations process.
We will make sure that the cost of ISO 13485 Certification in Kuwait is as minimal as possible. To get you ISO Certification Services in Kuwait than we assure you 100% guarantee results and we ensure that you will definitely be certified because have 100% success rates till date in getting our customers certified. So get in touch with us as early as possible and get your ISO 13485 certification in Kuwait at the earliest.
Mail us at contact@factocert.com for quick assistance.




















