ISO 13485 Certification
ISO 13485 certification is an industry specific standard. It specifies requirements for quality management system or QMS that can be used by an organization involved in one or more stages of the lifecycle of a medical device. The standard applies to component manufacturers and service organizations, as well as manufacturers of the finished devices.
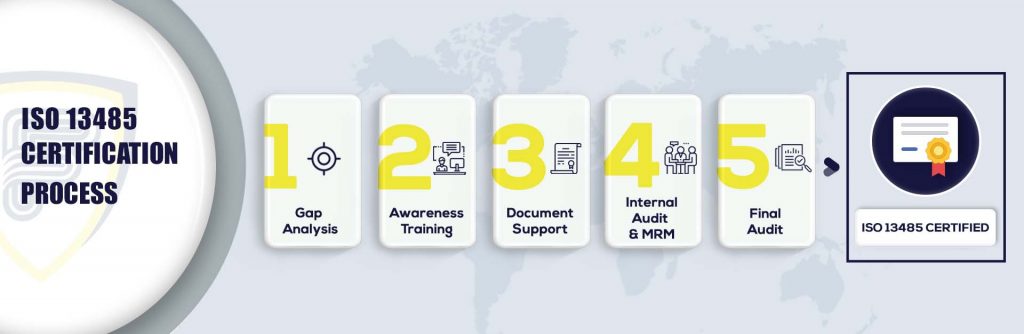
What are the steps to get ISO 13485 Certification?

Understanding the clause 8 of ISO 13485 – Measurement Analysis and Improvement
Firstly, you have to plan how your organization will monitor, measure and analyze processes to ensure product and QMS conformity and QMS effectiveness. You will have to establish methods to obtain and monitor customer feedback and processes to investigate complaints, review risk, take action and report results including to regulatory authorities where appropriate, your organization has to plan and conduct internal audits to determine whether the QMS is in conformity and processes are achieving plan results. You have to monitor and measure product during the manufacturing process to identify non-conforming product and take appropriate action to isolate such product and prevent unintended delivery or use. Lastly, it is essential to analyze data about your organization’s QMS, in order to estimate its suitability and efficiency and improve where indicated where appropriate you need to take corrective and preventive action in order to maintain QMS suitability, adequacy and effectiveness to ensure medical device safety and performance.
Benefits of being ISO 13485 certified:
- Helps minimize manufacturing defects safety as a key focus.
- Helps growth efficiency, cut costs and monitor performance.
- Determines that your company has the ability to produce safe medical devices.
Companies implementing ISO 13485 certification are expected to identify the regulatory requirements that apply to its activities under these rules:
Does the company have the responsibility and authority to recall product and what documentation is needed to be kept and for how long and finally a company implementing ISO 13485 is expected to incorporate the applicable regulatory requirements within the QMS, which means there should be procedures for reporting to regulatory, authorities and their customers and then they should be implemented procedures for traceability. Throughout ISO 13485 the phrase as appropriate is used, if your organization deems that a section of the standard does not apply. For example, your product does not have service or installation. You must note that as a justification as to why it does not apply, usually you state this in the scope and the quality manual.
For more Information : ISO 13485 Certification

























