ISO 13485 Certification In Qatar
ISO 13485 Certification in Qatar
ISO 13485 certification in Qatar is one of the highest standards which specify the requirement of a quality management system for the design and manufacturing process of medical devices. The pharmaceutical industries which want to demonstrate their methods that they are working very effectively and all the quality requirements are maintained up to the mark can certify by ISO 13485 standard. ISO 13485 certification in Qatar is one of the standards which is accepted worldwide, which gives equality requirement for all the medical manufacturing industries and helps to make all the regulatory and other requirements.
What are the steps to get ISO 13485 certification in Qatar?
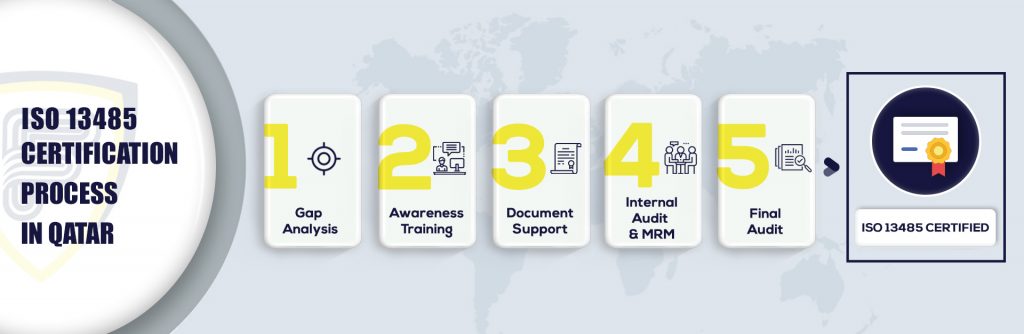

Why ISO 13485 certification in Qatar?
We know in this competitive market every organization preaches to get profit and urges for high-quality products. So one of the best remedies to overcome all these issues is by opting the implementation process of ISO 13485 registration services in Qatar and due to this reason the medical industries can make a profit and identify the risk and hazards that are occurring in the medical devices so that the end product or in the manufacturing process the defect can locate and high-quality products at the end.
ISO 13485 Certification in Qatar is one of the guaranteed & assurance where it involves in all the stages or phases of medical devices like manufacturing, installing, designing and assistance related to technical issues. It not only participates in that but also covers the post-production issues like distribution, services related to provision storage extra. ISO 13485 Certification audit in Qatar based on the quality management system standard ISO 9001, where it focuses more on the medical devices environment and processes.
If you certified by ISO 13485 certification in Qatar, it acts as one of the expansions in the market where you can gain a wide variety of opportunities if you have a good brand on your products which helps your organization to attract the customers. ISO 13485 certification consultants in Qatar are very flexible and to know ISO 13485 Certification cost in Qatar visit our website www.factocert.com
For the application of quality management system by industries or any organization There are several jurisdictions which have the requirements related to regulatory name the organization which wants to opt ISO 13485 certification in Qatar to their organization expects specific terms and let us see what are they.
- The applicable regulatory requirements would be identified and organizations roles and responsibility would be specified
- For certain activities, it identifies the regulatory requirements and applies to it by using specific functions.
- Along with the quality management system, these regulatory requirements should be incorporated that are applicable.
The regulatory requirements differ from Nation to Nation, country to country and region to region. And it is the responsibility of the organization to understand how the statutory requirements would play an essential role by national standards which apply to the medical devices. ISO 13485 certification cost in Qatar is beneficial.
More about ISO 13485 certification in Qatar:
ISO 13485 Certification in Qatar is a kind of standard which can be used by external parties, private parties, certification bodies, customers and other requirements which are related to the right things.
The framework of the implementation process of an organization is influenced by these terms
- The varying needs of the organization
- Objectives mainly associated with the organization.
- The products which are provided by the organization
- Structure and size of the organization
- Legal or regulatory requirements which apply to the activities carried out in the organization.
- And it also depends upon the environment of the organization, the changes in the background or any other influences that might affect the organization’s environment so that the conformity of the medical devices is concerned. To know how to get ISO 13485 certification in Qatar, our company would assist you
Requirements of ISO 13485 certification in Qatar:
Scope: In this section, ISO 13485 audits in Qatar speaks about the standard and how it can apply to the organization
- The process approach importance
- Product and services regulatory requirements have to include.
- To have continual improvement process has to be in place
Normative reference: Along with ISO 13485 Certification in Qatar, standard specific requirements and recommendations from ISO 9000 should use. ISO 13485 audit services in Qatar provide a Framework for the quality management system vocabularies and fundamentals.
Terms and definitions: In this section, different types of definitions used in the standards listed, which includes ISO 9001:
- Medical device which is active implantable
- Active medical device
- Notice related to advisory
- Complaint related to customers
- Medical devices which are implantable
- Labeling
- Medical device
- Sterile medical device
Sections 4 to 8 are the requirements of the ISO 13485 Certification in Qatar, which has to be made by your organization who wants to be certified organization of ISO 13485. ISO 13485 Certification consultants in Qatar helps to achieve all the risk and to know more services contact us at contact@factocert.com
General requirements of ISO 13485 certification in Qatar:
For the overall quality management system, this section gives a condition
- Requirements for the documentation for purpose includes
- Corporate quality management system along with quality manual
- Procedures that are required
- Forms and records which are needed
- Document control
- Control of forms


Responsibility of Management:
This section gives a requirement for the top management to take the roles and responsibilities in implementing the quality management system
- Responsibility of the management
- Objectives and policies related to quality
- Customer satisfaction and focus of customer
- Management review
Our services:
ISO 9001 Certification
ISO 9001: ISO 9001 certification Is among those basic and basic international standard published by the international organization for standardization for setting up the quality demands within an organization which focuses on continuous improvement and customer satisfaction and are irrespective of size, kind, or scope.
The ISO 9001 certification Specifies regarding quality management systems (QMS) is a set of standards that helps organizations ensure they satisfy the customer and other stakeholders’ requirements inside statutory and regulatory requirements related to your good or service. ISO 9001 deals with the fundamentals of QMS, ISO 9001 deals with all the requirements that organizations wanting to satisfy the standard needs to fulfill.
ISO 14001 Certification
ISO 14001: ISO 14001 certification Provides the essential guidelines and tools for your organization to handle its environmental performances. And it’s come to be one of the mandatory factors for the organization to do business with others. It can help to demonstrate that the organization accepts responsibility for environmental concerns and meet all kinds of legal duties that are employed by businesses.
ISO 14001 standards associated with environmental Management system and this standard help organizations to reduce their operations, procedures that negatively impact the environment comply with applicable laws, regulations, and other environmentally oriented conditions; and always improve to protect the environment such as water, air, or land.
ISO 45001 Certification
ISO 45001: ISO 45001 certification Is among those major business tools enabling the necessity for occupational health and safety management system that protects the employees and other visitors from work-related accidents, injuries, and disease. This could be one of the boosting factors for the employees at the organization due to which they can work very efficiently and productively with no threats.
ISO 45001 is an ISO standard for management systems of occupational health and safety (OH&S), published in March 2018. The objective of ISO 45001 is that the decrease occupational injuries and ailments, such as promoting and protecting physical and mental health.
The standard is based on OHSAS 18001, Conventions and recommendations of the International Labour Organization including ILO OSH 2001, and federal standards. It has elements that are added to BS OHSAS 18001 that is replacing over a three-year migration interval from 2018 to 2021.
ISO 22000 Certification
ISO 22000: ISO 22000 certification Is one of the industry-specific standards that is been created by ISO providing the requirements for a food safety management system that can be put on the food distribution chain, beginning from farm to fork. It functions as a highly effective marketing tool that showcases the quality & safety variables of food products.
Contrasts between HACCP and ISO 22000
- ISO 22000 is the prerequisites for food a safety management system that’s fully based on quality principles.
- HACCP is a risk management tool that prevents food safety hazards from ever occurring in the first location. It addresses the risks from assessing and controlling the chemical, biological and physical hazards.
ISO 20000-1: ISO 20000-1 certification Is an international standard that fulfills the needs for information technology service management systems.
ISO/IEC 20000 is the first international standard for service management. ISO/IEC 20000 was originally designed to reflect best practice advice contained within the ITIL framework, though it both supports additional IT service management frameworks and procedures such as Microsoft Operations Framework and components of ISACA’s COBIT framework.
ISO/IEC 20000-1 specifies requirements for “setting, implementing, maintaining and continually enhancing a service management system (SMS). An SMS supports the management of this service lifecycle, including the preparation, design, transition, delivery, and improvement of services, which meet agreed requirements and deliver value for customers, users and the organization providing the services.”
ISO 31000 Certification
ISO 31000: ISO 31000 Certification is a standard designed for supplying the requirements for risk management.
ISO 31000 was published as a standard in 2009 and supplies a Standard about the implementation of risk management. The goal of ISO 31000 would be to be more applicable and adaptable for”any person, private or community enterprise, institution, group or individual.” ISO 31000 is not designed for a particular business category, management system, or subject matter subject in mind, rather provides a best-practice arrangement and guidance to all operations involved with risk management.
ISO 31000 seeks to Offer a globally recognized standard for Practitioners and companies employing risk management procedures to replace the array of present standards, methodologies, and paradigms that contrasts between businesses, subject topics, and areas. For this function, the recommendations provided in ISO 31000 may be customized to some organizations.
ISO 10002 Certification
ISO 10002: ISO 10002 certification Is associated with customer satisfaction especially in regards to handling complaints. Having a set of guidelines that are supplied by ISO 10002, alternatives are given to boost the organization’s performance.
Customer Satisfaction is understood to be a measurement that determines how happy customers are using a firm’s goods, services, and abilities. Customer satisfaction information, such as polls and ratings, can help a company determine the way to best enhance or changes its goods and services. It’s a measure of how products and services provided by a company meet or surpass customer expectations.
ISO 27001 Certification
ISO 27001: ISO 27001 certification is among those important international standards that Assist the industries to fulfill all the Privacy legislation by supplying the demands of information security management systems. By getting the execution procedure of ISMS, the organizations can attest they are reliable collaboration partners.
ISO/IEC 27001 is an international standard on how to manage information security. The standard was initially published together by the International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC). It details requirements for establishing, implementing, maintaining, and continually improving an information security management system (ISMS) — the aim of which is to help organizations make the information resources they maintain more secure. Organizations that meet the standard’s requirements can choose to get certified by an accredited certification body after the successful conclusion of a scheduled appointment.
Information Security management system (ISMS) defines and handles controls an organization needs to implement to ensure it is sensibly protecting the confidentiality, availability, and integrity of resources from threats and vulnerabilities. The core of ISMS includes information risk management, a process that involves the assessment of the risks, an organization must cope with the management and security of assets
ISO 17025 Certification
ISO/IEC 17025: ISO 17025 certification is a Special standard that has an International recognition that defining the need for the competence of testing and calibration laboratories.
ISO/IEC 17025 is the standard for which most labs must hold Certification to be deemed technically competent. In many cases, suppliers and regulatory authorities will not accept test or calibration results from a lab that isn’t accredited.
ISO/IEC 17025 was issued with the International Organization for Standardization. There are several commonalities with the ISO 9000 standard, but ISO/IEC 17025 is much more unique in demands for competence and employs directly to those organizations that make testing and calibration results and is based on a bit more technical fundamentals. Laboratories utilize ISO/IEC 17025 to implement a quality system targeted at improving their ability to consistently produce valid results.
ISO 22301 Certification
ISO 22301: ISO 22301 Certification is an international standard that is set to supply the needs for a business continuity management system.
ISO 22301 certification defines requirements to Plan, establish, implement, manage, monitor, review, maintain, and always improve a registered management system to protect against, reduce the probability of the event and prepare for, respond to, and recover from disruptive incidents if they arise.
It applies to all organizations regardless of the type, size, And nature of this organization. Organizations that employ a business continuity management system (BCMS) dependent on the requirements of ISO 22301 can undergo a formal assessment procedure through which they can obtain accredited certification from this standard.
ISO 50001 Certification
ISO 50001: ISO 50001 Certification is a sole standard that has worldwide recognition for providing the prerequisites for an energy management system.
Energy management contains the preparation and performance of energy production and energy consumption components as well as energy distribution and storage.
The standard specifies the requirements for creating, implementing, maintaining, and improving an energy management system, whose goal is to enable an organization to adhere to a systematic strategy in achieving continual improvement of energy performance, including energy efficiency, energy security, energy usage, and consumption.
The energy management system can also Refer to a system made to achieve energy efficiency through process optimization by simply reporting granular energy use by individual pieces of equipment.
ISO 29990 Certification
ISO 29990: ISO 29990 certification is one of the most followed standards By non-formal education institutes and training centers.
The ISO 29990 was published in an ISO Standard in 2010,” ISO 29990″Learning services for training and additional education – Basic requirements for service providers”. The structure of ISO 29990 maps both service demands and management conditions.
ISO 21001 places requirements on a management system. Both, additionally, replace ISO 29990 in a considerably enhanced form but do not need to be used together. Depending on the interests of the user of this standard, they may be combined or used separately.
ISO 13485 Certification
ISO 13485: ISO 31485 Certification in Oman is an International Standard, that specifies the Quality management systems that are, involved with medical devices.
ISO 13485 represents the prerequisites for a comprehensive ISO 31485 Certification in Oman quality Management system for the design and fabrication of medical devices.
CE mark Certification
CE mark: CE mark certification and ISO 13485 Certification in Oman for A product can be marketed from the European Union Area without breaking any lawful conditions. CE marking indicates the safety, health, and environmental protection representation of the item.
HALAL Certification
HALAL: HALAL certification and ISO 13485 Certification in Oman is mandated for many countries. Halal describes this food product, which is Devised according to regulations and regulations of Islamic that governs what is lawful, permissible, approved, legal, licit, permitted, and tidy
HACCP Certification
HACCP: HACCP certification and ISO 13485 Certification in Oman is a Management system standard that offers the need for hazard analysis critical control points. It addresses the risks by analyzing and controlling the chemical, biological and physical hazards.
GMP Certification
GMP: GMP certification and ISO 13485 Certification in Oman is important. Good manufacturing practice isn’t anything but measures that are accepted to have a controlled practice according to quality standards.
GLP Certification
GLP: GLP certification and ISO 13485 Certification in Oman is necessary. Good laboratory principle is one of the systematic methods of communicating and conditioning the environment of a laboratory by preparation, performing, observation, recording, and reporting the outcomes.
SA 8000 Certification
SA 8000: SA 8000 Certification and ISO 13485 Certification in Oman is a certifiable international standard that motivates an organization to employ, build and maintain good social practices in the workplace.
VAPT Certification
VAPT: Vulnerability assessment And penetration testing and ISO 13485 Certification in Oman are the two kinds of tests consisting of different strength and total vulnerability assessment. Any cybersecurity exposures could be recognized with the help of both of these security testing methods.
CMMI Certification
CMMI: The capability maturity Model integration and ISO 13485 Certification in Oman is a powerful means of reducing the risks in software solutions And other services created from the organization.
Advantages of ISO 13485 certification in Qatar:
- ISO 13485 certification bodies in Qatar is one of the standards which is recognized globally and has become one of the models of quality management system standard, which is related to medical industries.
- If ISO 13485 consultant services certify you in Qatar gives assurance to your customers that you are following one of the best quality standards, which makes our organization more trustworthy And reliable.
- ISO 13485 consulting services in Qatar not only helps to meet the customer satisfaction and invoice the continual improvement, but it helps you to meet all the legal requirements, regulatory requirements, marketing and contractual requirements.
- ISO 13485 consultancy in Qatar helps you to provide the safest products to the end-users without any harm.
- ISO 13485 consultancy services in Qatar acts as one of the cost-saving factors by reducing the defects and accidents of the product, and the process is carried out in the organization.
- ISO 13485 certification in Qatar help to identify all the risks and provide an effective risk management system.
- ISO 13485 Certification in Qatar also involved in the activities such as increasing the efficiency of the employees, process and services.
These are the benefits of ISO 13485 certification in Qatar the quality management system for medical devices which helps the organizations which are related medical industries and helps to provide the effective management system by involving all the requirements according to the standard.
Our Services
Our Clients










































ISO 13485 audit services in Qatar are essential because if you’re facing the ISO 13485 audit in Qatar you have to be very careful about every parameter in your organization but when you join hands with Factocert. We as an ISO 13485 Consultancy Service provider in Qatar , are tagged up with so many different ISO certification bodies in Qatar .
Benefits of ISO 13485 Certification in Qatar
You will have the edge over your list of ISO 13485 certified companies in Qatar.
Marketing becomes very easy when you have certification tagged on to your name and which will help you to get into a global market and be a global player.
Employee satisfaction rates increases which are directly proportional to your employee retention and by retaining your critical employees you will have higher stability as a company
A government will recognize you for having such ISO 13485 certification in Qatar
You will be automatically qualifying for any tenders because most of the companies require you to certify for ISO 13485 certification in Qatar for participating in tender
Customer satisfaction rates will go high, and you will not have to face any more consequences from your customers or your vendors
Are you looking for
ISO 13485 Certification Consultants in Qatar
What else are you waiting for the only step you have to take care about is getting in touch with us, and we would take the best care, and in no time you would be certified for the relevant ISO 13485 certification in Qatar you prefer. Most thing companies worry about is ISO 13485 service cost in Qatar but let us just tell you that ISO 13485 certification in Qatar Cost is not what you should be thinking of because when this certification can give a boost to your organizations process.
We will make sure that the cost of ISO 13485 in Qatar is as minimal as possible. To get you ISO 13485 Certification Services in Qatar than we assure you 100% guarantee results and we ensure that you will definitely be certified because have 100% success rates till date in getting our customers certified. So get in touch with us as early as possible and get your ISO 13485 certification in Qatar at the earliest.
Mail us at contact@factocert.com for quick assistance.




















