GMP Certification In Ghana
ISO Certification In Ghana
GMP Certification In Ghana, Factocert is one of the leading GMP Certification providers in Ghana. We provide Halal Consultant service in Accra, Kumasi, Tamale, Cape Coast, Ashaiman, and other major cities.Great production practice (GMP) is a system for ensuring that products are continually produced and also controlled according to high-quality standards. It was developed to decrease the risks associated in any pharmaceutical production that can not be eliminated by examining the final product in Ghana. The primary dangers are:
- Unforeseen contamination of products, causing damages to wellness or even death.
- Inaccurate labels on containers suggest that clients receive the incorrect medicine.
- Insufficient or too much energetic ingredients cause inadequate treatment or unfavorable impacts in Ghana.
GMP covers all aspects of production, from the beginning materials, facilities and devices to the training and also personal health of staff. Comprehensive, written treatments are vital for every procedure that might influence the quality of the finished product. There have to be systems to give documented evidence that the right procedures are regularly followed at each action in the production procedure – each time a product is made in Ghana. That has established in-depth standards and complete production methods. Many nations have developed their demands for GMP based upon WHO GMP. Others have integrated their needs, for example, in the Association of South-East Asian Nations (ASEAN), in the European Union and also through the Drug Examination Convention in Ghana.
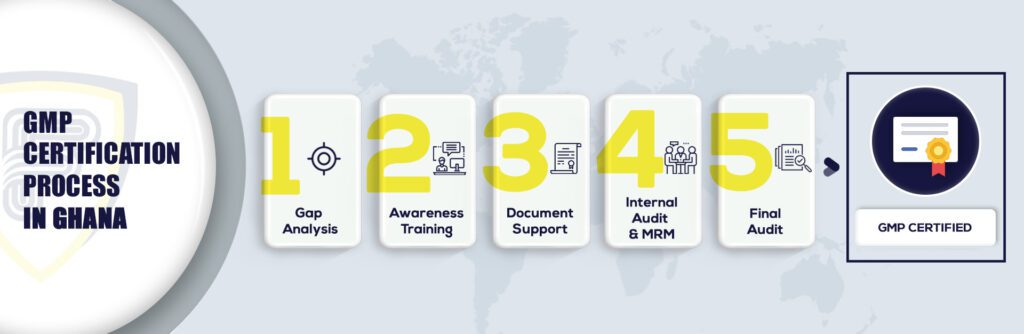
What are the steps to get GMP Certification in Ghana?

Advantages of GMP Certification in Ghana
- GMP Certification brings immense benefits to your organization. A few essential advantages are:-.
- Boosts the food security administration system.
- Boosts customer confidence in your products.
- It helps reduce operating costs because of rework and penalties for non-compliance.
- It helps increase export possibilities.
- Lowered replication of examinations.
- Cost saving.
- One of the primary advantages for the manufacturer is incredibly improved top-quality systems and top-quality agreements at the excipient supplier.
- We have seen these renovations in the months leading up to GMP Certification and continue during the years, promptly complying with GMP Certification.
- Some excipient mass manufacturers were currently at a high GMP conformity and Certification level.
- GMP Services in Ghana Improves the food safety administration system.
- GMP Consultants in Ghana Boosts Customers’ rely on your products.
- GMP Certification Cost in Ghana Aids to reduce operating expenses due to remodeling and also charges because of non-compliance.
- GMP in Ghana Helps boost export chances. Decreased replication of inspections.
Demands for GMP Certification in Ghana:
- Good documentation makes up a needed part of the quality control system. The composed policy protects against mistakes arising from spoken communication, and clear paperwork authorizes the tracing of tasks done.
- Kind has to be developed, prepared, examined, and distributed with treatment.
- Records need to be approved, authorized, and dated by the appropriate skilled and authorized individuals.
- Records must have unique components. The title, nature, and also function should be stated. They must be laid out in a well-ordered style and be easy to inspect; the reproduced papers should be clear and legible.
- Documents have to be consistently reviewed and maintained and upgraded. When a document has been revised, systems must be run to avoid accidental use of superseded documents (For instance, only current documents must be accessible for usage).
- Documents must not be transcribed; nonetheless, documents call for information access; these entries may be made in easy-to-understand, readable handwriting utilizing a suitable enduring medium (i.e., not a pencil). Sufficient space must be provided for such entrances.
The following GMP Certification in Accra method problem ‘paperwork and documents’ may be helpful for pharmaceutical suppliers to meet the presumption of different regulative companies. Compose excellent treatments, follow them, and assume what has happened in an office if procedures are not readily available. Individuals depend on even more elderly staff members to tell them exactly how to make points and afterward do their job. This is phenomenal for an organization making garden pots; however not so good when the products being made are pharmaceuticals and can even trigger fatality! Find out the task before you begin writing the procedure.
Produce a brief collision of the vital actions and key points related to the job. Following treatments, it is all very well to have a great written plan of action in position; however, to guarantee a regulated and suitable efficiency, they must be complied with; it is a GMP requirement. Frequently, the steps described in a written policy might need to show up to the most effective method of functioning, and taking faster ways might conserve time.
These are the two primary reasons for this:
- Many faster ways might produce a risk that can be costly in the end.
- Each step in treatment has been covered for a function.
Keep good documents; Good records allow one to track all activities performed throughout batch manufacture, from obtaining basic materials to the end product release; they generate a background of the set and its distribution.
Which industries are eligible for GMP Certification in Ghana?
- Production Companies.
- Pharmaceutical Firms.
- Food Manufacturing Business.
Exactly how to obtain GMP Consulting Providers in Ghana?
Factocert is providing GMP Consulting Services in Ghana with comprehensive knowledge and experience in all Global food and pharmaceutical items Requirements.
In addition to performing the execution or review of international standards, Factocert offers third-party auditing solutions and certification of international standards. Decide wisely and select a consulting organization like Factocert that will always work from the client’s perspective and ensure they are delighted with our providers. Please visit our website at www.factocert.com to learn more about our services. We would be pleased to help you with any issues you may be having.
For More Information Do Visit: www.factocert.com
ISO 9001 is an internationally recognized identification mark for qualifying criteria of quality administration. The International Standard Organization, that is recognized by over 120 countries as the jurisdiction in establishing quality standards for products and services.
ISO 13485 Certification is for Quality control systems for medical devices is an International Organization for Standardization, whose goal would be to set quality criteria for pharmaceutical, biotechnological and generic drug products. The main objective of ISO is to establish quality standards for drugs, generic drugs, and surgical materials and equipment that are used by health care and industrial industries in the production of medicines, devices, and equipment. The company places quality goals, quality control steps, quality management systems, performance data specifications, and labeling or packaging requirements. This international standard was accepted by various authorities worldwide. As of the writing of this article, ISO has 27 member states and is expanding rapidly in every area of the pharmaceuticals business.
ISO 14001 is the international Standard for developing and implementing a hazardous waste management program. ISO 14001 was established in 1996 to help ease the compliance of resources of hazardous wastes. Its purpose is to establish a uniform system of technology rules and guidelines necessary for handling such wastes. The standard is always updated through consensus, which makes it the most comprehensive international standard on toxic wastes. ISO 14001 is the international standard for layout and implementing a hazardous waste management program
ISO/IEC 17025 General requirements for the competence of testing And calibration laboratories is your principal ISO standard used by testing and calibration labs. In most nations, ISO/IEC 17025 is the norm for which most labs need to hold certification in order to be deemed technically competent. Oftentimes, providers and regulatory authorities will not accept test or calibration results from a laboratory that isn’t accredited.
The International Standard Organization (ISO) is the sole authority for establishing quality parameters for products. It sets the superior specifications used to ascertain what can and can’t be offered in the market. If a food or beverage company wishes to sell its products in almost any country across the world, they need to be accredited to do so by the ISO. If you’re a producer who wants to market food items or beverages beyond your country, it’s vital that you comply with each of the ISO 22000 regulations. Not doing this could lead to heavy fines, penalties, and loss of your small business.
ISO/IEC 27001 is an International standard about how best to handle information security. The standard was originally published together by the International Organization for Standardization (ISO) and the International Electrotechnical Commission (IEC) in 2005[1] and then revised in 2013. [two ] It details requirements for establishing, implementing, maintaining, and constantly improving an information security management system (ISMS) — the aim of which is to help organizations create the data resources they hold more protected. [3] A European upgrade of this standard was printed in 2017. [4] Organizations that meet the standard’s requirements can opt to be certified by an accredited certification body following successful completion of an audit.
ISO 45001, an Internationally recognized International Standard Organization (ISO) standard For occupational health and safety management systems, has been created to Address problems regarding occupational health. The International Standard Organization, which is a subdivision of the US National Committee for Standards Is a not-for-profit organization that sets quality standards for goods and services. An organization that has achieved the Maximum standing of “IEC 60 Annex 1” is an ISO member.
Our Services
Our Clients










































GMP audit services in Ghana are essential because if you’re facing the GMP auditor in Ghana you have to be very careful about every parameter in your organization but when you join hands with Factocert. We as an GMP Consultancy provider in Ghana, are tagged up with so many different GMP certification bodies in Ghana.
Benefits of GMP Certification in Ghana
You will have the edge over your list of GMP certified companies in Ghana.
Marketing becomes very easy when you have GMP certification in Ghana tagged on to your name and which will help you to get into a global market and be a global player.
Employee satisfaction rates increases which are directly proportional to your employee retention and by retaining your critical employees you will have higher stability as a company
A government will recognize you for having such GMP certification in Ghana
You will be automatically qualifying for any tenders because most of the companies require you to certify for GMP certification in Ghana for participating in tender
Customer satisfaction rates will go high, and you will not have to face any more consequences from your customers or your vendors
Are you looking for
GMP Certification Consultants in Ghana
What else are you waiting for the only step you have to take care of is getting in touch with us, and we would take the best care, and in no time you would be certified for relevant GMP certification in Ghana you prefer. Most thing companies worry about is GMP service cost in Ghana but let us just tell you that Halal Consultant in Ghana is not what you should be thinking of because when this certification can give a boost to your organizations’ process.
We will make sure that the cost of GMP Certification in Ghana is as minimal as possible. To get you GMP Certification Services in Ghana than we assure you 100% guarantee results and we ensure that you will definitely be certified because have 100% success rates to date in getting our customers certified. So get in touch with us as early as possible and get your GMP certification in Ghana at the earliest.
Mail us at contact@factocert.com for quick assistance.
Frequently Asked Questions
What is ISO Certification in Ghana?
The ISO Certification in Oman stands for International Organization for Standardization. It plays an essential role in maintaining various market sectors’ standards. It starts right from manufacturing an item to providing a product. It is an independent, international organization that develops standards for ensuring the safety, quality, and efficiency of the services and products across Ghana cities like Accra, Kumasi, Tamale, Cape Coast, Ashaiman, and other major cities.
Who Needs ISO Certification in Ghana?
For industries in Ghana, ISO certification might be called for by legislation or contractually. But, even if that’s not the situation, satisfying ISO criteria has many advantages for organizations: Saving money and time by recognizing and resolving persisting issues, Improving system, and process effectiveness.
What are the types of ISO Certifications mandatory in Ghana?
While all the ISO Standards are necessary for different organizations, some of the mandatory ISO Certification Standards in Ghana are:
- ISO 9001 Certification: Quality Management System
- ISO 14001 Certification: Environmental Management System
- ISO 45001 Certification: Occupational Health and Safety
- ISO 27001 Certification: Information Security Management System
- ISO 22000 Certification: Food Safety Management System
What is the cost of ISO Certification In Ghana?
Although the cost of ISO Certification in Ghana depends on the type of ISO Standards, Factocert provides the best ISO Certification services at the most affordable price across Ghana.




















