ISO 13485 Certification In Australia
ISO 13485 Certification In Australia
ISO 13485 Certification In Australia, Factocert is one of the leading ISO Certification providers in Australia. We provide ISO Consultant service in Sydney, Melbourn, Brisbane, Perth, Adelaide, and other major cities. ISO 13485 Certification in Australia is all about preserving quality Management system for medical devices
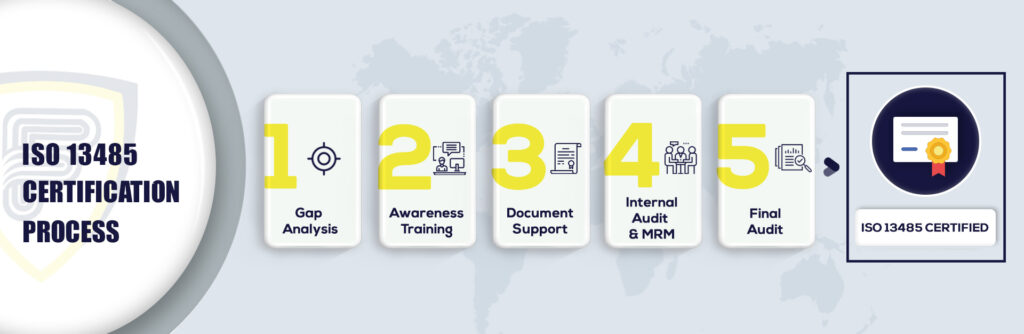
5 Steps to Get ISO 13485 Certification in Australia:

More about ISO 13485 Auditors in Australia:
ISO 13485 Auditors in Australia standard for the organization is a tactical choice and the execution and design of the organization’s quality management depend upon specific variables and they’re:
- The environment of this organization changes from this environment, and the Power of the organizational environment has resulted in the conformity of medical devices
- Varying demands of the organization
- Organizations goals that are special
- The Goods that are being supplied from the organization
- Worker Procedure for this organization
- Organizational size and structure of ISO 13485 Certification Cost in Australia.
- Legal requirements which are related to these organizational actions
The requirements for ISO 13485 Auditors in Australia given by Standard are pertinent to this organization are no matter the size and kind. Particular requirements which are defined by ISO 13485 that are pertinent to this organization but it isn’t performed by the organization would be the sole duty of the organization. And it’s the duty of those organization’s quality management systems for controlling, monitoring and maintaining the procedures. The standard employed by your organization will probably demonstrate to the buyers and users of the healthcare devices which you’ve fulfilled both their needs alongside the federal requirements. That’s known worldwide hence you’re able to find a more variety of chances around the world.
ISO 13485 Certification Consultants in Australia
Slimming down a Strategy to generate ISO 13485 Certification Consultants in Australia a layout is a fundamental step to come up with a prosperous medical device procedure. Medical businesses must undergo numerous bargains to be able to generate a regulation, security product, and also a safe item. Implementation of ISO 13485 standard within an organization will assist the management system to look and produce a procedure for the healthcare devices. Along with regard to particular requirements in the customers, the organization will have the ability to satisfy the client’s needs using a proper medical apparatus management system.
Preparation of Policies during the first stages to make a nice infrastructure and structure of this organization will maintain roles and obligations in the best management system.
Establishment Activities to cut back the very ideal strategy has become easily the most essential crucial measure of execution of any International standard.
When the program is Prepared the next stage begins with the development and design of a management system program. This specific stage aids the organization in identifying aims and objectives. Allocating time and handling risk activities to finish the entire job will help to finish the stage.
The Plan must Be of equality in which it must fulfill the customer requirement. A few of the expectations in the customers in the medical sector can easily be attained with the support of both all standards. Risk management and RISC healing processes employed by the management. Investigation of present market values connected to medical devices and other costing things have to be discussed one of the best management staff. Considering all the appropriate study making a valid and statutory necessity in the front of the management system can help to diminish the very ideal design.
The results of This input have to be determined with the assistance of the management system. The output is made of raw materials particles which must fulfill the comprehensive item. Identification of the solution and understanding the manufacturing businesses. Inspection has to be performed before conducting some experiments about the meeting. Manufacturing procedures and other crucial specifications connected to surroundings have to be understood before demonstrating the procedure.
When the layout Is prepared it’s very crucial to examine it in front of the client before going together using the meeting. The display has to be achieved with environmentally friendly components and materials together with ensuring that the facility is secure to make sure all of the adequate pursuits. Verification of the two outputs and input has to be ready before designing the procedure.
Any modifications in The procedure have to be revealed with the aid of flow identification and a diagram of this system has to be put. With the support of advice supplied from ISO 13485 standard and complete layout and development for a management system could be created.
ISO 13485 Certification Bodies in Australia
ISO 13485 acts as a benchmark for The businesses that take part in medical manufacturing. The ISO Consultant service in Sydney, Melbourn, Brisbane, Perth, Adelaide ISO 13485 Certification Bodies in Australia organizations that have experienced or attained ISO 13485 Certification Cost in Australia standard have enhanced in their own goods quality, regulatory compliance, ISO 13485 Certification Cost inn Australia and reliability and therefore are compared with the business best practices. Organizations of any kind or dimensions which are involved with developing and implementing ISO 13485 matches quality management system. Therefore, the long run of this quality management system, as well as your organization, Depends on your current decision, ISO 13485 Certification Cost in Australia
ISO 13485 Certification Cost in Australia
- ISO Certification Cost in Australia assists the organization to have a constant advancement in their civilization.
- It helps the organization to get a better quality of those devices that will Lead to equity
- Among the best way to have client satisfaction in a greater degree
- It helps to Decrease the Price and increase efficiency by with better quality variables
- Decisions are based upon the info’s and details which are coordinated with the organizational strategic aims.
ISO 9001 Certification
ISO 9001 Sets out the criteria for a quality management system. It may be employed by any organization, big or small, irrespective of its field of action. In reality, there are over one million companies and organizations in over 170 countries certified to ISO 9001.
ISO 9001 needs to Demonstrate its ability to consistently provide products and services that meet customer and applicable statutory and regulatory demands, and this standard aims to improve customer satisfaction through the successful use of this system,
such as processes for improvement of this system and the assurance of conformity to customer and applicable regulatory and regulatory conditions.
ISO 14001 Certification
ISO 14001 Sets out the criteria for an environmental management system and can be accredited to. It maps out a framework that a business or organization can follow to set up a successful environmental management system.
ISO 14001 helps an organization achieve the intended consequences of its environmental management system, which Offers value for the environment, the organization itself, and Interested Parties.
ISO 45001 Certification
ISO 45001 ISO 45001 specifies requirements for the Occupational health and safety (OH&S) management system, and gives guidance for its use, to allow organizations to supply safe and healthy workplaces by preventing work-related injury and ill health, as well as by proactively improving its OH&S performance.
A lot of people died from work-related accidents or diseases. That is An ISO committee of occupational health & safety experts place to work to develop an International Standard using the capability to save employees in the office so ISO introduced ISO 45001 standard.
ISO 22000 Certification
ISO 22000 sets out the prerequisites for a food safety management system. It stands out exactly what an organization needs to do to demonstrate its ability to control food safety hazards to ensure that food remains safe.
It can be used by any organization irrespective of its size or location from the food chain.
ISO/IEC 20000-1: it’s information technology – service management system (SMS) standard. It specifies requirements for your service provider to plan, establishes, implement, operate, monitor, review, claims, and improve an SMS.
The requirements include the plan, transition, shipping, and enhancement of services to meet agreed-on service demands.
ISO 31000 Certification
ISO 31000, Risk management — Guidelines, provides fundamentals, A framework, and a process for managing risk. It can be employed by any organization regardless of its size, action, or business. ISO 31000 provides a degree of reassurance in terms of financial resilience, professional standing, and environmental and safety results.
ISO 31000 can be used Throughout the life of this organization and may be applied to virtually any activity, including decision-making at all levels.
ISO 27001 Certification
ISO/IEC 27001 standards Are the work of joint ISO and IEC (International Electrotechnical Commission) the technical committee, Information security, cybersecurity, and private security.
ISO/IEC 27001 is broadly Known, supplying prerequisites for an information security management system (ISMS), though there are over just several dozen standards in the ISO/IEC 27000 household.
Applying them empowers organizations of any sort to control the security of assets such as financial information, intellectual property, employee information, or information secured by third parties.
ISO 17025 Certification
ISO/IEC 17025 specifies The overall requirements for the competence of testing and calibration laboratories.
ISO/IEC 17025 enables laboratories to show that they function competently And generate valid results, thereby boosting confidence in their work both nationally and around the world.
In Addition, it helps facilitate collaboration between laboratories and Other bodies by generating wider acceptance of outcomes between states. Evaluation reports and certificates can be taken from 1 country to another without the need for additional testing, which, in turn, improves international trade.
ISO 50001 Certification
ISO 50001 Designed to support organizations in all sectors, this ISO standard provides a practical way to improve energy use, through the development of an energy management system (EnMS).
This relies on the management system model of continual improvement also used for additional renowned standards such as ISO 9001 or ISO 14001. This also makes it much easier for organizations to integrate energy management in their overall efforts to enhance quality and environmental management.
CMMI Certification
CMMI: Capability Maturity Model Integration (CMMI) and ISO 9001 Certification in Oman is a process-level improvement training and appraisal program. Administered by the CMMI Institute, it was developed at Carnegie Mellon University (CMU). It is needed by many U.S. Government contracts, particularly in program development.
CMMI can be used to guide process development across a job, division, or entire organization. CMMI defines the Subsequent maturity levels for processes: First, Managed, Defined, Quantitatively Managed, and Optimizing.
ISO 29990 Certification
ISO 29990: specifies basic requirements for providers of learning services within non-formal education and training.
ISO 13485 Certification
ISO 13485: ISO 13485 specifies requirements for a quality management system for the medical devices where an organization needs to demonstrate its ability to provide medical devices.
Such organizations can be involved in one or more stages of their Life-cycle, including design and development, manufacturing, storage and supply, installation, or servicing of a medical apparatus, and design and development or provision of associated activities.
ISO 13485:2016 can also be used by suppliers or external parties that offer a product, including quality management system-related services to such organizations.
ISO 13485 is designed to be used by organizations included in The design, manufacturing, installation, and servicing of medical devices and related services. It can also be used by internal and external parties, such as certification bodies, to help them with their auditing processes.
CE Mark Certification
CE Mark The Conformitè Europëenne (CE) Mark and ISO 9001 Certification in Oman has described as the European Union’s (EU) mandatory conformity marking for controlling the goods marketed in the European Economic Area (EEA) as of 1985.
The CE marking signifies a manufacturer’s declaration that products comply with the EU’s New Approach Directives.
These directives not only apply to products within the EU but also to products that are manufactured in or designed to be sold at the EEA. This produces the CE marking familiar worldwide even to those unfamiliar with the EEA.
There is an EU requirement that goods not in conformity with all the terms of the directives aren’t permitted to circulate into the territories of the member countries
CE marking doesn’t provide any particular information to the consumer. It’s not a quality assurance statement, but it doesn’t reveal evidence of third-party testing, and it should not be mistaken with any certification mark of the type issued by international or European notified exam bodies.
Certain directives include a choice for the Responsible organization to present a statement of conformity saying a product fulfills the requirements of the related directives.
Halal Certification
Halal is a word Designating any object or an action that’s permissible to use or engage in, based on Islamic law. It’s the reverse of haram. And the ISO 9001 Certification in Oman term is employed to designate food seen as permissible by Islamic law.
Halal is the Arabic word meaning lawful or permitted. About food, it is the dietary standard, as prescribed in the Qur’an (the Muslim scripture).
These conditions are commonly used in terms of Food goods, meat products, cosmetics, healthcare products, pharmaceuticals, food ingredients, and food contact materials.
When many things are halal or haram, some things are not apparent. Further information is needed to categorize them as halal or haram.
HACCP Certification
HACCP Hazard analysis and critical control factors, or HACCP and ISO 9001 Certification in Oman is a systematic preventive Strategy To food safety by biological, compound, bodily hazards, and more recently radiological dangers in manufacturing processes which could cause the final product to be dangerous and layouts measures to reduce these risks to a safe level.
In this manner, HACCP tries to prevent hazards instead of trying to inspect finished products for the consequences of these hazards.
The HACCP system can be used in Any Way stages of A food series, from food production and preparation procedures including packaging, distribution, etc…
HACCP and ISO 9001 Certification in Oman was recognized internationally as a logical tool for adapting conventional review methods into a contemporary, science-based, food safety system.
According to risk assessment, HACCP plans permit both industry and authorities to devote their funds effectively by building and auditing safe food manufacturing practices.
GMP Certification
GMP: Good Manufacturing Practice (GMP) and ISO 9001 Certification in Oman is a system for ensuring that products are consistently produced and controlled following quality standards.
It’s designed to minimize the risks involved in any pharmaceutical manufacturing that must not be eliminated throughout testing the final product.
GLP Certification
GLP: The OECD Principles of Good Laboratory Practice (GLP) and ISO 9001 Certification in Oman ensure the creation of high-quality and reliable test information linked to the safety of industrial chemical compounds and preparations.
SA 8000 Certification
SA 8000: The SA8000 and ISO 9001 Certification in Oman Standard is the world’s foremost social certification program. The SA8000 Standard and Certification System provides a platform for organizations of all kinds, in virtually any market, and in almost any country to conduct business in a means that is reasonable and decent for employees and to show their adherence to the highest social standards.
Made by SAI in 1997 as the first credible social certification, it has led the industry for over 20 decades.
VAPT Certification
VAPT: Vulnerability Assessment and Penetration Testing (VAPT) and ISO 9001 Certification in Oman are equally security services that focus on identifying vulnerabilities in the network server, and system infrastructure.
The two services serve another purpose and are carried out to attain different but complementary goals.
Why choose Factocert for ISO 13485 Certification in Australia?
Here is a piece of brief information regarding the documents required while implementing the ISO 13485 standard. Get the ISO 13485 certification cost in Australia just by getting in touch with us at contact@factocert.com by involving the operational procedures, recovery plans, and corrective actions the standard would be more effective when it’s implemented in your organization and know more about ISO 13485 cost in Australia.
For more information: ISO 13485 Certification in Australia.
Our Services
Our Clients










































ISO 13485 auditors in Australia and services in Australia are essential because if you’re facing the ISO 13485 auditors in Australia you have to be very careful about every parameter in your organization but when you join hands with Factocert. We as an ISO 13485 Certification Consultants in Australia, are tagged up with so many different ISO 13485 certification bodies in Australia
Benefits of ISO 13485 Certification in Australia
You will have the edge over your list of ISO 13485 certification companies in Australia.
Marketing becomes very easy when you have ISO 13485 certification in Australia tagged on to your name and which will help you to get into a global market and be a global player.
Employee satisfaction rates increases which are directly proportional to your employee retention and by retaining your critical employees you will have higher stability as a company
A government will recognize you for having such ISO 13485 certifications
You will be automatically qualifying for any tenders because most of the companies require you to certify for ISO 13485 certification in Australia for participating in the tender
Customer satisfaction rates will go high, and you will not have to face any more consequences from your customers or your vendors
Are you looking for
ISO 13485 Certification Consultants in Australia
What else are you waiting for the only step you have to take care about is getting in touch with us, ISO 13485 Certification Bodies In Australia and we would take the best care, and in no time you would be certified for relevant ISO 13485 certification in Australia and ISO 13485 Certification Bodies In Australia you prefer.
Most things companies worry about is ISO 13485 Certification cost in Australia but let us just tell you that ISO 13485 Certification cost in Australia is not what you should be thinking of because when this certification can give a boost to your organizations process.
We will make sure that the ISO 13485 certification cost in Australia is as minimal as possible, ISO 13485 Auditors In Australia. To get you ISO 13485 Certification consultants in Australia than we assure you 100% guarantee results and we ensure that you will definitely be certified because have 100% success rates to date in getting our customers certified, ISO 13485 Auditors In Australia. So get in touch with us as early as possible and get your ISO 13485 certification in Australia at the earliest, ISO 13485 Auditors In Australia.
Mail us at contact@factocert.com for quick assistance.




















