GLP Certification In Turkey
GLP Certification In Turkey
GLP Certification in Turkey or the Good Laboratory Practices includes the methods for observing, documenting, archiving, and reporting environmental and health safety investigations for implementation.
GLP Certification refers to laboratory activity testing specific goods in people and animals. These items are standard:
- Food Supplements
- Medical Supplies
- Cosmetic Items
- Substances Used in the Prevention and Control of Harmful Organisms
- Feed Supplements
- Manufacturing Materials
In a GLP-compliant laboratory, test operations are coordinated and controlled, documented and reported correctly. Organizations eligible for the Good Laboratory Practices Certificate include:
- Laboratories that undertake safety research on non-clinical medication manufacture.
- Laboratories engaged in the development of biocidal products
- Agricultural pesticide research laboratories
- Laboratories that operate with dangerous chemical compounds
- Food additive research laboratories
- Cosmetic product development laboratories
Factocert is the leading GLP Consultant in Turkey, providing GLP Certification in Ankara, Edirne, Eskisehir, Bursa, Mardin, Istanbul, and other major cities in Turkey with services such as implementation, documentation, audit, training, gap analysis, and registration process at an affordable cost.
What Are The Steps To Get GLP Certification In Turkey?
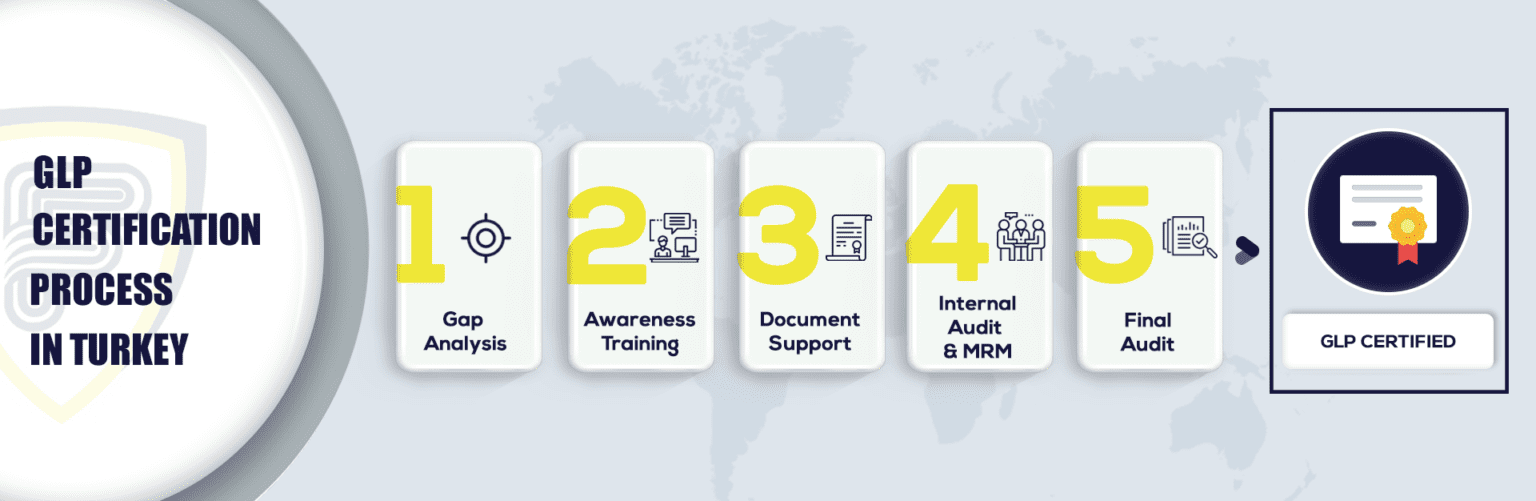

More About GLP Certification in Turkey:
Good Laboratory Practice Certification in Turkey (GLP) is a well-known acronym for laboratory quality. GLP is a formal regulation formulated in 1978 by the US Food and Drug Administration (FDA) that registered pharmaceutical manufacturers; however, the primary target of the regulation was not the chromatography laboratory or even the analytical laboratory.
GLP Certification in Turkey governs non-clinical laboratory safety (i.e., animal toxicity testing) to ensure that novel molecular entities developed by pharmaceutical companies are safe before being administered to human volunteers and patients in clinical trials.
As the regulation’s title indicates, the goal of Good Laboratory Practice Certification in Turkey has not changed since it was published: Good Laboratory Practice for Nonclinical Laboratories.
While it is true that the Laboratory in GLP in Turkey contains chromatography, the regulation’s overall goal does not. The significant laboratories impacted by the legislation were histology, clinical chemistry, and pathology, utilized to examine toxicological samples.
In addition to FDA standards, the Organization for Economic Cooperation and Development (OECD) has produced GLP regulations, which have the same broad objective as FDA regulations but with specific variances.
GLP Certification Principles in Turkey:
The GLP Certification Principles in Eskisehir aim to accelerate the generation of high-quality test data. The related quality of test data serves as the foundation for data acceptability across nations. Duplicate testing may be avoided if relevant nations can confidently depend on test data generated in other countries, saving time and resources. The use of GLP Certification in Ankara should assist in avoiding the establishment of technical trade barriers and promote the protection of human health and the environment. Some of the Principles are:
- Responsibilities of Test Facility Management
- Quality Control Program
- Facilities
- Instruments, materials, and reagents
- Test Equipment
- Test and Reference Materials
- SOPs (Standard Operating Procedures)
- The Study’s Execution
- Study Results Reporting
- Record and material storage and retention
Many firms of various sorts, including Testing & Calibration Companies and Medical Laboratory Companies, use GLP Certification in Mardin as a most recognized means of offering client pride and managing product and service quality within their chosen industry.
GLP may be utilized not only to provide advancements and ensure quality, but it is also considered a guarantee of product and service consistency across borders, languages, and cultural divides. As a result, getting GLP certification is advantageous in terms of reputation and practicality. Implementing GLP Certification in Istanbul is a fresh approach to company assurance. GLP Services in Turkey promotes client satisfaction by analyzing its objectives and goals so that GLP Consultants can implement GLP for all industries in Turkey.
What role does GLP Consulting Services play in Turkey?
GLP Certification registration in Turkey is not restricted to chemicals. GLP Certification Services in Turkey also addresses food additives, medical equipment, colour additives, food packaging, and other non-pharmaceutical items or components.
One of the primary goals of GLP Consultants in Ankara is to oversee what scientists perform and how they do safety/quality testing of chemical and biochemical goods and pharmaceutical development.
GLP Certification in Bursa is also necessary for registration and licencing for medicines, food additives, veterinary medication products, insecticides, and specific bio-products. GLP Certification Consultant in Turkey attempts to demonstrate that no safety or quality data has been falsified or modified in any manner.
GLP Certification in Edirne also ensures regulators and research Commands that the data and material acknowledged are based on study outcomes and conclusions. Only in this manner will they be able to proceed and finish the necessary assessments.
GLP in Turkey requires analysts to preserve adequate, accurate, valid, consistent, and correct records of each testing procedure, technique, and methodology to reduce false claims or other differences.
What Are The Advantages Of Effective Laboratory Practice?
- A clear flow of traceability with a precise trail from document production through retention/deletion
- Increased confidence in the quality and dependability of laboratory data
- Improved ability to provide the appropriate results the first time
- The increased overall productivity of laboratory operations via the GLP system
- Improved brand reputation as a result of efficient laboratory management
- Less demand for rework
- Significantly reduced time spent examining non-revenue producing activities/cases
What Are The Requirements For GLP Certification in Turkey?
GLP comprises two critical components:
- First, GLP is a Quality Management System that specifies a set of quality requirements for research design, data collecting, and results reporting.
- The second critical component of the GLP system is quality assurance (QA). The QA unit is an impartial internal authority that validates and monitors all stated rules and procedures throughout the study’s conduct, analysis, and reporting.
Some of the most critical requirements in the GLP implementation process are:
- The OECD GLP Principles
- Resource management in terms of human and physical infrastructure.
- Characterization is all about gaining information. Characterization in non-clinical investigations is concerned with the test item (typically a chemical substance) and the test method (often a live animal). GLP needs at least these two entities to be characterized.
Why Should You Choose Factocert for GLP Certification in Turkey?
Factocert is the leading GLP Consultant in Turkey, offering GLP Certification in Turkey with services such as implementation, audit services, certification, documentation, gap analysis, and registration. GLP registration in Turkey is precise, fast, and affordable. You have many options for doing so, including contacting us at contact@factocert.com or visiting www.factocert.com . For more information visit GLP Certification in Turkey
Our Services
Our Clients










































GLP audit services in Turkey are essential because if you’re facing the GLP audit in Turkey you have to be very careful about every parameter in your organization but when you join hands with Factocert. We as a GLP Consultancy Service provider in Turkey, are tagged up with so many different GLP certification bodies in Turkey.
Benefits of GMP Certification in Turkey
You will have the edge over your list of GLP certified companies in Turkey.
Marketing becomes very easy when you have certification tagged on to your name and which will help you to get into a global market and be a global player.
Employee satisfaction rates increases which are directly proportional to your employee retention and by retaining your critical employees you will have higher stability as a company
A government will recognize you for having such GLP Certification in Turkey
You will be automatically qualifying for any tenders because most of the companies require you to certify for GLP Certification in Turkey for participating in tender
Customer satisfaction rates will go high, and you will not have to face any more consequences from your customers or your vendors
Are you looking for
GLP Certification Consultants in Turkey
What else are you waiting for the only step you have to take care about is getting in touch with us, and we would take the best care, and in no time you would be certified for relevant GLP certification in Turkey you prefer. Most thing companies worry about is GLP service cost in Turkey but let us just tell you that GLP cost in Turkey is not what you should be thinking of because when this certification can give a boost to your organizations process. We will make sure that the cost of GLP in Turkey is as minimal as possible.
To get you GLP Certification Services in Turkey than we assure you 100% guarantee results and we ensure that you will definitely be certified because have 100% success rates till date in getting our customers certified. So get in touch with us as early as possible and get your GLP certificate in Turkey at the earliest.
Mail us at contact@factocert.com for quick assistance.




















