ISO 13485 Certification In Brazil
ISO 13485 Certification In Brazil, Factocert is one of the leading ISO 13485 Certification providers in Brazil. We provide ISO 13485 Consultant service in Sao Paulo, Rio de Janeiro, Manaus, Salvador, Brasilia, and other major cities.
ISO 13485 Certification Brazil is one of the Stand-alone standards that’s been published by the international organization for standardization to define the requirements for the quality control system of the associations that are involved with medical device manufacturing.
What are the steps to get ISO 13485 Certification in Brazil?
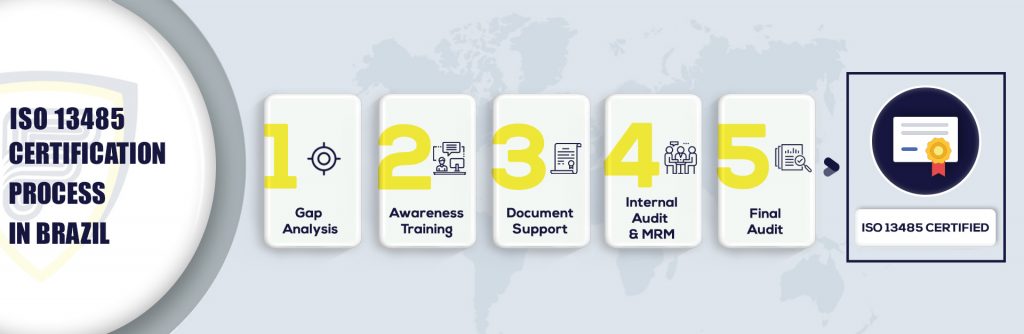

ISO 13485 is based on the internationally recognized standard ISO 9001 and it incorporates the requirements that are related to medical devices procedure. The standard was established to assist the Medical device organizations to set up a quality control system to demonstrate its consistent development and layout, storage creation, installation, distribution, decommissioning preservation, and disposal of all devices related to medical sectors alongside the layout and development and other associated tasks.
The standard ISO 13485 Certification in Brazil mainly focuses on The process approach towards quality management within an organization. This processing system is a reviewed based on the sequence such as, for the inputs and inputs and additional interactions of the procedures.
Any activities that receive the input signal and Deliver the output are thought to be a procedure. And, in most situations, the outcome of one activity is that the input of the following activity and so forth. The quality system of the standard should identify and eradicate the risk and specifically like meeting the customer and regulatory requirements.
- The organization that makes use of a process approach to quality management leads to
- Better consistency and understanding in surpassing on meeting the product demands
- Describe each process and provides a value-added perspective
- It is helpful to achieve functionality to a higher degree.
- Depending upon the performance information the continual advancement of the approach is done and not on the opinion of specific speculations.
What’s the Overall requirement of the ISO 13485 Certification in Brazil?
- Quality management system in medical devices should go with the process that holds internationally good across the world.
- Establishing the policies concerning quality along with other medical industry-specific add the first stage.
- Moving into the files the differences involving the international standard has to be satisfied with the help of a consultant.
- Meeting the essential documents to have been organized construction in medical businesses is assured.
- Return policies and putting them in a highly effective manner from the direction system will improvise every action in the sections.
- Each of the systems has to be tracked and processed to obtain expected results.
Advantages of ISO 13485 Certification in Brazil
ISO 13485 standard acts as a standard for The industries which are involved in medical manufacturing. The organizations that have undergone or attained ISO 13485 Certification cost in Brazil standards have improved in their product quality, regulatory compliance, and reliability and are about the industry best practices. Organizations of any type or size that are Involved in implementing and developing ISO 13485 matches quality management system. Hence, the future of this ISO 13485 Consultants in Brazil (quality management system-Medical devices) and about your organization depends upon your present choice
Now We’ll see few other benefits of ISO 13485 Certification in Brazil
- It enables the company to have a constant advancement in their civilization.
- It helps the organization to have an improved quality of the apparatus which will result in equity
- Among the best solution to have client satisfaction at a higher level
- It helps to reduce the Price and increase efficiency by having better quality factors
- Decisions are based on the Info’s and facts that are aligned with The organizational strategic goals
For more details visit: www.factocert.com or contact@factocert.com
Our Services
Our Clients










































ISO 13485 audit services in Brazil are essential because if you’re facing the ISO 13485 audit in Brazil you have to be very careful about every parameter in your organization but when you join hands with Factocert. We as an ISO 13485 Consultancy Service provider in Brazil, are tagged up with so many different ISO 13485 certification bodies in Brazil.
Benefits of ISO 13485 Certification in Brazil
You will have the edge over your list of ISO 13485 certified companies in Brazil.
Marketing becomes very easy when you have ISO 13485 certification in Brazil tagged on to your name and which will help you to get into a global market and be a global player.
Employee satisfaction rates increases which are directly proportional to your employee retention and by retaining your critical employees you will have higher stability as a company
A government will recognize you for having such certifications
You will be automatically qualifying for any tenders because most of the companies require you to certify for ISO 13485 certification in Brazil for participating in tender
Customer satisfaction rates will go high, and you will not have to face any more consequences from your customers or your vendors
Are you looking for
ISO 13485 Certification Consultants in Brazil
What else are you waiting for the only step you have to take care of is getting in touch with us, and we would take the best care, and in no time you would be certified for relevant ISO 13485 certification in Brazil you prefer. Most thing companies worry about is ISO 13485 service cost in Brazil but let us just tell you that ISO 13485 cost in Brazil is not what you should be thinking of because when this certification can give a boost to your organizations process.
We will make sure that the cost of ISO 13485 in Brazil is as minimal as possible. To get you ISO 13485 Certification Services in Brazil than we assure you 100% guarantee results and we ensure that you will definitely be certified because have 100% success rates till date in getting our customers certified. So get in touch with us as early as possible and get your ISO 13485 certificate in Brazil at the earliest.
Mail us at contact@factocert.com for quick assistance.




















